Top Class Actions’s website and social media posts use affiliate links. If you make a purchase using such links, we may receive a commission, but it will not result in any additional charges to you. Please review our Affiliate Link Disclosure for more information.
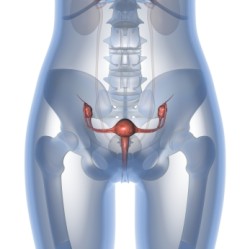
Carlaw first had the Mirena IUD inserted in 2009 with no initial complications. At first, the Mirena IUD caused no complications to the plaintiff, and she used it unaware that she was susceptible dangerous side effects.
However, in April 2010 the plaintiff had begun experiencing painful symptoms and went for a medical examination. It was then she was told the Mirena IUD had perforated and migrated from her uterus. On April 22, 2011, Carlaw underwent laparoscopic surgery to have the device removed because it had been embedded on the outside of plaintiff’s uterine cavity.
Carlaw is suing Bayer alleging the Mirena IUD label was insufficient and made no mention of these risks. Bayer failed to point out any recent or relevant studies, as well as any related FDA notifications made to the public. Carlaw states that she never would have used the product if she had known about the possibility of perforation. She also said that Bayer had the obligation to warn her and other patients of these possibilities, because they are relying on the information given at the time.
Carlaw accused Bayer Pharmaceuticals of negligence, false advertising, concealing information and misrepresenting a product.
Overview of Mirena IUD Complications
The Mirena IUD was first approved by the FDA in December 2000, and is manufactured by Bayer Pharmaceuticals. It is the first intrauterine device approved and sold in the United States, gaining more 2 million users since its release. It is a small t-shaped device, made of biologically safe plastic that can be inserted into the body. It is directly implanted into the uterus, and releases a small dose of leveonorgestrel directly into the bloodstream to prevent ovulation and sperm-to-egg fertilization. IUDs are among the most popular forms of birth control in the United States, but have recently been associated with troubling health complications.
As mentioned earlier, at the device’s endpoint is a hormone called levonorgestrel, which is a progestin hormone directly released into the bloodstream in the uterine lining. Once it reacts with the body’s biological functions it prevents sperm-to-egg fertilization as well as ovulation. Ideally, these devices would last up to five years, and give minimal complications to the patient.
If the IUD perforates the uterus it could migrate into a different area of the patient’s body. Once the device migrates, there’s a chance it could embed itself onto a different internal area, and cause problems. The problems reported from include cramping, bleeding, the need for surgical removal, organ embedment, and ectopic pregnancies.
The Mirena IUD lawsuit is In re: Mirena IUD Products Liability Litigation, MDL No. 2434, in the U.S. District Court for the District of Minnesota.
Join a Free Mirena Class Action Lawsuit Investigation
If you or a loved one had the Mirena IUD inserted after January 1, 2000 and had to have surgery – or will be required to have surgery – to remove the IUD because it migrated, you may be eligible to take legal action against the manufacturer. Joining a Mirena class action lawsuit or filing an individual Mirena IUD lawsuit may help you recover compensation for medical bills, pain and suffering and other damages. Obtain a free case evaluation now:
Oops! We could not locate your form.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2024 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.














