Top Class Actions’s website and social media posts use affiliate links. If you make a purchase using such links, we may receive a commission, but it will not result in any additional charges to you. Please review our Affiliate Link Disclosure for more information.
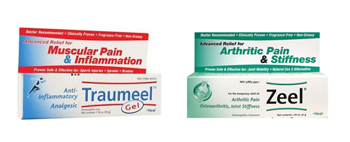
Under the terms of the class action settlement, Heel will reimburse consumers who seek refunds for their homeopathic product purchases, up to a limit of $150 per customer. The company also agreed to clarify the science behind its drugs that claim to treat a variety of ailments including joint pain, allergies and skin conditions.
Plaintiff Robert A. Mason filed the class action lawsuit on December 21, 2012. He claims that he was deceived about the nature of the homeopathic products with respect to the dilution of the products’ active ingredients, their lack of effectiveness, and Heel’s claims that the products were “’natural,’ ‘clinically proven,’ and ‘doctor recommended.’”
Mason claims that Heel’s assertions are misleading and violate the Unfair Competition Law, False Advertising Law, Consumer Legal Remedies Act and the Magnuson-Moss Warranty Act. He also asserts claims of unjust enrichment and breach of express and implied warranties.
Mason brought the Heel class action lawsuit on behalf of a nationwide class of consumers who purchased homeopathic products made by Heel since December 21, 2008. He specifically targeted Heel’s Traumeel and Zeel treatments, claiming that the products did not perform as advertised. Mason claims that the active ingredients in these products were diluted so severely that it was likely that no molecule derived from the original “extract” was actually included in the final product. He alleges that claims of the products’ effectiveness were founded on biased studies.
In February, Heel filed a motion to dismiss the class action lawsuit, alleging that Mason simply attacked homeopathy and that his lawsuit was preempted by the Federal Food, Drug and Cosmetic Act, which preempts deceptive advertising cases involving homeopathic drugs. However, the company withdrew its motion before a judge could issue a ruling.
As part of the class action settlement, Heel has agreed to place a disclaimer on its packaging stating: “These statements have not been reviewed by the Food and Drug Administration. They are supported by traditional homeopathic principles.”
This disclaimer will be included in all of the company’s advertising that make a claim related to the products’ usage. Heel will also include a claim about the level of dilution of the active ingredients included in the products and will refrain from using the term “natural” unless the product contains all natural ingredients. The company agreed to stop using the words “Clinically Proven,” “Proven … Effective,” “Doctor Recommended” and “[Used] by doctors worldwide” unless the company relies on independently researched clinical studies or discloses the percentage of homeopathic doctors and allopathic doctors who recommend the product.
Details on how to file a claim to receive compensation from the Heel Homeopathic Product Class Action Settlement were not immediately available. Keep checking Top Class Actions or sign up for our free weekly e-newsletter below to receive settlement updates.
The plaintiff is represented by Ronald A. Marron, Syke Resendes and Alexis M. Wood of the Law Offices of Ronald A. Marron APLC.
The case Robert A. Mason v. Heel Inc., Case No. 3:12-cv-03056, in the U.S. District Court for the Southern District of California.
UPDATE 1: A California federal judge granted preliminary approval to the Heel Traumeel, Zeel Class Action Settlement on Oct. 30, 2013.
UPDATE 2: Details on how to file a claim for the Heel homeopathic products class action settlement are up! See how to file a claim to receive up to $150 here.
UPDATE 3: A federal judge approved the Mason v. Heel class action settlement on March 13, 2014. The Settlement Administrator says it expects to begin distribution of settlement funds in late July 2014. If you get a payment, let us know!
UPDATE 4: Payments from the Heel homeopathic class action settlement were mailed the second week of November. Learn more: http://topclassactions.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2024 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.













19 thoughts onHeel Homeopathic Company Reaches $1M Class Action Settlement
I GOT MY CHECK FOR $45.24 TODAY. YAY!!! THANK GOD AND T.C.A.
UPDATE 4: Payments from the Heel homeopathic class action settlement were mailed the second week of November. Learn more: http://topclassactions.com/lawsuit-settlements/lawsuit-news/43855-turbotax-heel-homeopathic-class-action-settlements-pay/
THANKS. T.C.A. FOR THE UPDATE.
UPDATE: A federal judge approved the Mason v. Heel class action settlement on March 13, 2014. The Settlement Administrator says it expects to begin distribution of settlement funds in late July 2014. If you get a payment, let us know!
Robert A. Mason is obviously working as a toady of the pharmaceutical industry to discredit natural medicine, or he is just a money grubbing law-suit parasite. Homeopathy has been successfully used for over a hundred years. The Heel company makes fantastic products! Maybe they embellished their advertising claims, but that doesn’t warrant a malicious attack. Now thanks to Robert A Mason, the Heel company is leaving the USA and Canada and their excellent products will only be available overseas. Any one tagging along on this lawsuit is also an ignorant money grubbing parasite. Hooray for the American “if it moves, sue it” legal mentality. Another good company down the drain.
Any homeopathic remedy will not work if the consumer exposes themselves to any strong oil or fragrance such as a cough candy with peppermint and/or eucalyptus, tobacco, menthol, oil of oregano, muscle balms such as A535 or tiger balm, or even coffee. Remedies also do not work if you choose the incorrect remedy for your problem. To expect homeopathic results while indulging in, or continuing toxic habits,(smoking or eating a typical unhealthy north american diet) will also minimize successful results. There is more to the health puzzle than just what is printed on a label. As for the minute amounts of active ingredients in the product, that is the principle of Homeopathy. Educate yourself. Nobody ever died from a homeopathic remedy.
Need to file a claim as have used Allergy products
How can I participate ?
MAY I FILE A CIVIL COMPLAINT AGAINST HEEL’S homeopathic products, AS THEY DO NOT PERFORM AS ADVERTISED…
Need to participate