Top Class Actions’s website and social media posts use affiliate links. If you make a purchase using such links, we may receive a commission, but it will not result in any additional charges to you. Please review our Affiliate Link Disclosure for more information.
Studies Show Tygacil Complications May Be Fatal
By Amanda Antell
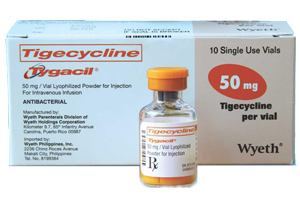
Tygacil (tigecycline) is typically administered intravenously, and should be administered with a medical practitioner present. While the black box warning came in 2013, there had been an earlier warning about Tygacil from the U.S. Food & Drug Administration (FDA) in September 2010. The FDA announced that it had analyzed clinical data from Tygacil injury reports and found that most associated deaths stemmed from progression of the patient’s infection. Furthermore, there were a significantly higher number of deaths in patients who had hospital-acquired pneumonia. The current black box warning label reflects this risk, as well as mentioning that patients who were administered Tygacil had a higher risk of death when compared to other drugs.
The 2013 announcement was based off a meta-analysis consisting of ten studies, which showed increased mortality among patients who had received Tygacil. According to these reports, out of 2,640 patients who had received Tygacil, 2.5 percent of the patients died, compared with 1.8 percent of 2,628 patients who were given other antibacterial drugs.
Several drug safety advocate groups have petitioned for the black box warning since the FDA’s warning in 2010 and have criticized the FDA for its latent action. Due to the belated action of the black box warning label, there are hundreds of Tygacil lawsuits being filed against Pfizer, accusing the company of negligence, misrepresenting a product and concealing information.
Overview of Tygacil Complications
As mentioned before, Tygacil is a prescription antibiotic medication that is used to treat complex skin and abdominal infections. The drug was first was approved by the FDA in 2005, and is the first drug to be approved from the new drug family, the glycylcyclines. Tygacil disrupts the protein synthesis in the bacterial cells, which inhibits growth and reproduction, eventually causing the bacteria to die out because it cannot reproduce. This mechanism contrasts greatly with traditional antibiotics, which normally kill the bacteria.
When Tygacil was initially released, it was given first-priority release review by the FDA because it had shown its antibacterial strength through clinical trials. The drug’s main point of difference was that it could attack infections that were resistant to most other antibiotics. Tygacil proved to be a popular choice among patients, and reached sales of more than $152 million in 2012.
File a Tygacil Lawsuit Today
If you believe that you or a loved one have been the victim of a Tygacil injury, you have legal options. Please visit the Tygacil and Tigecycline Class Action Lawsuit Investigation. There, you can submit your claim for a free legal review and if it qualifies for legal action, a seasoned Tygacil lawyer will contact you for a free, no-obligation consultation. You will be guided through the litigation process at no out-of-pocket expenses or hidden fees. The Tygacil injury attorneys working this investigation do not get paid until you do.
All class action and lawsuit news updates are listed in the Lawsuit News section of Top Class Actions